As at 23 March 2021, Swissmedic, in collaboration with the regional pharmacovigilance centres (in particular with the Ticino EOC reference centre), has evaluated 862 reports of suspected adverse drug reactions (ADR) to COVID-19 vaccinations in Switzerland. The reports corroborate the side effects profile identified during the authorisation studies and described in the medicinal product information. There is no evidence to date of any new safety signals, so the positive ratio of benefits to risks of the vaccines currently in use is still the same.
862 reports of suspected adverse reactions to COVID-19 vaccines in Switzerland evaluated
26.03.2021
460 reports involve Pfizer/BioNTech's Comirnaty®, while 393 are associated with Moderna's COVID-19 vaccine. In nine cases, the vaccine was not specified. A small number of reports explicitly describe reactions following the second dose of the vaccine (n=49 for Comirnaty, n=21 for Moderna). Most of the reports were submitted by medical professionals while 57 (6.6%) came directly from those affected, i.e. the patients.
According to information from the FOPH, around 1.25 million vaccine doses have been administered in Switzerland up to and including 21 March 2021, and approximately 466,000 people have been fully vaccinated. Since there is often a delay in sending reports on adverse drug reactions to Swissmedic and it also takes time to carefully evaluate them, it is not possible to directly compare the reports shown here with the vaccinations given. Retrospectively, however, there is an approximate reporting rate of one per 1,000 doses administered.
The majority of the reports concerned women (67.9%). Men were affected in 28.2% of cases, and there were a few cases (3.9%) where no gender was specified.
The average age of those affected was 64.4 years (range 16–101), with 42.7% aged 75 or over. In the cases classified as severe (n = 263) the average age was 70.6 years, and for reports temporally linked to a death (n = 37) it was 83.8 years.
At 599 (69.5%) most of the reports were classified as not serious, while 263 reports (30.5%) were classified as serious. In these latter cases, the people affected were either treated in hospital or their reactions were classified as medically significant for other reasons. In most cases, however, the persons concerned were not in danger.
The majority of the reports involved more than one reaction (a total of 2,154 reactions in 862 reports, equating to an average of 2.5 reactions per report).
The reactions most commonly reported in the cases classified as serious were fever (38), shortness of breath (25), hypersensitivity (19) /anaphylactic reactions (9), infection with COVID-19 disease (16)1, vomiting (12), dizziness (12), headache/migraine (17) and reactivation of shingles (8). Since a single report generally involves more than one reaction, reactions such as fever or headache are reported in cases that are classified as serious overall as well as in non-serious cases.
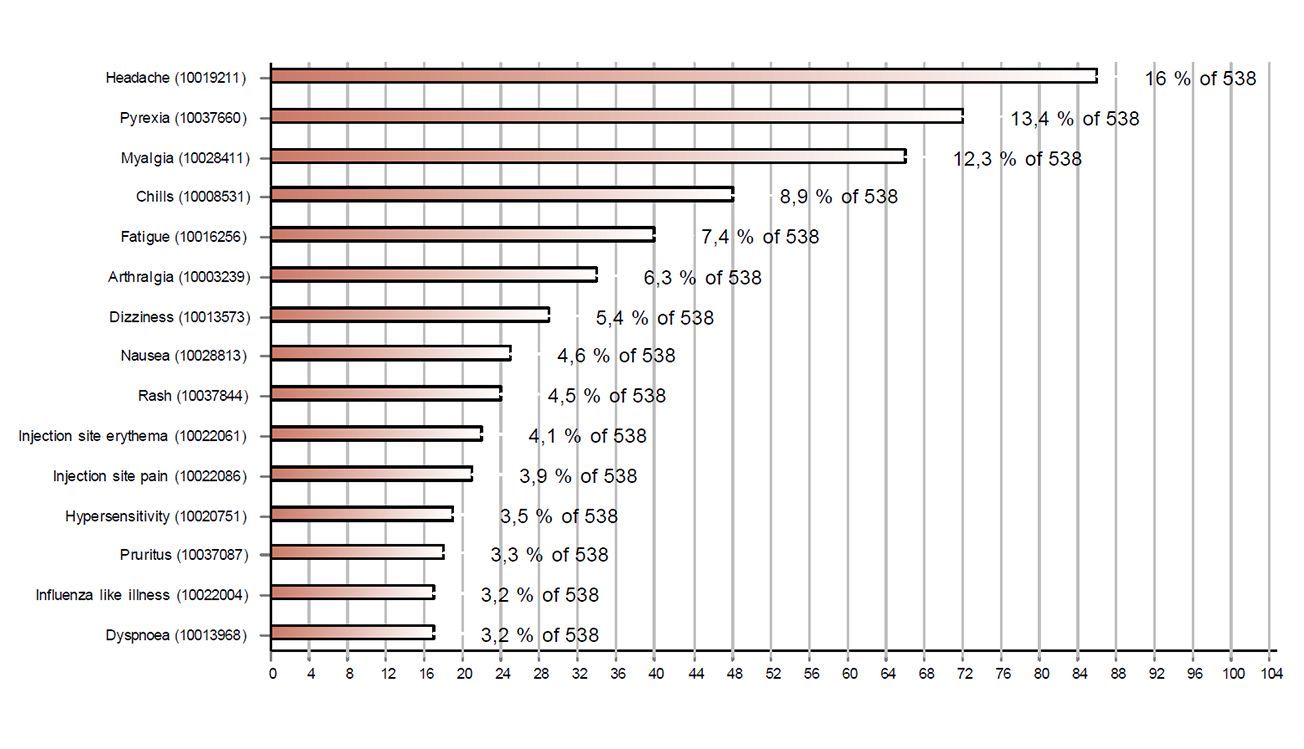
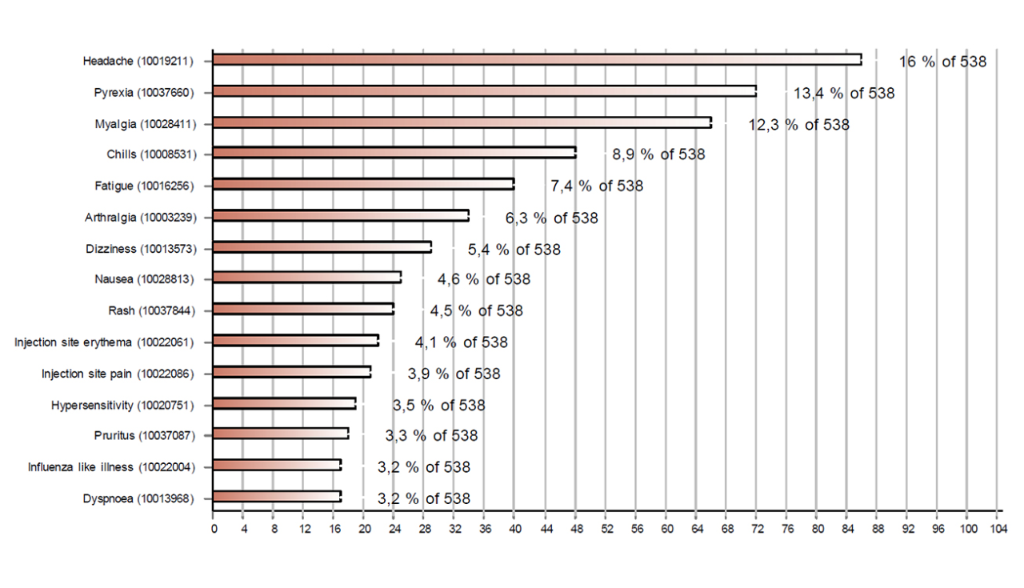
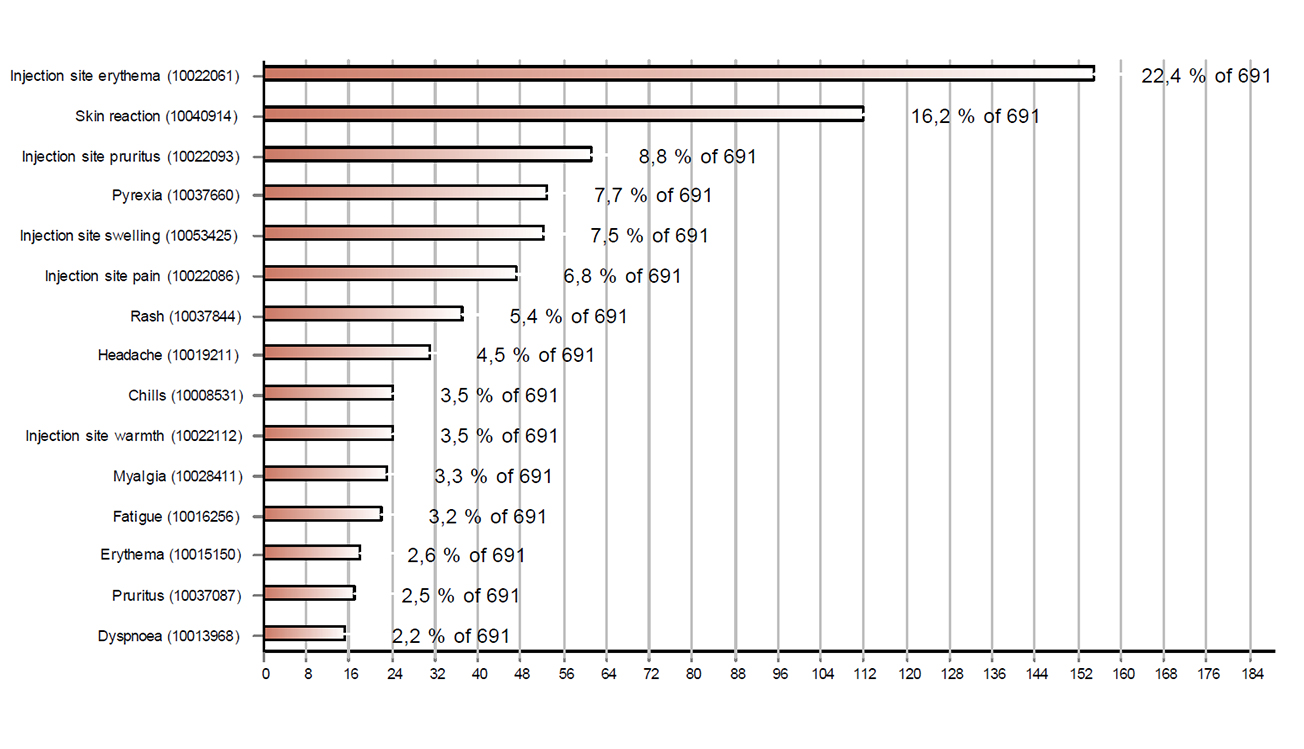
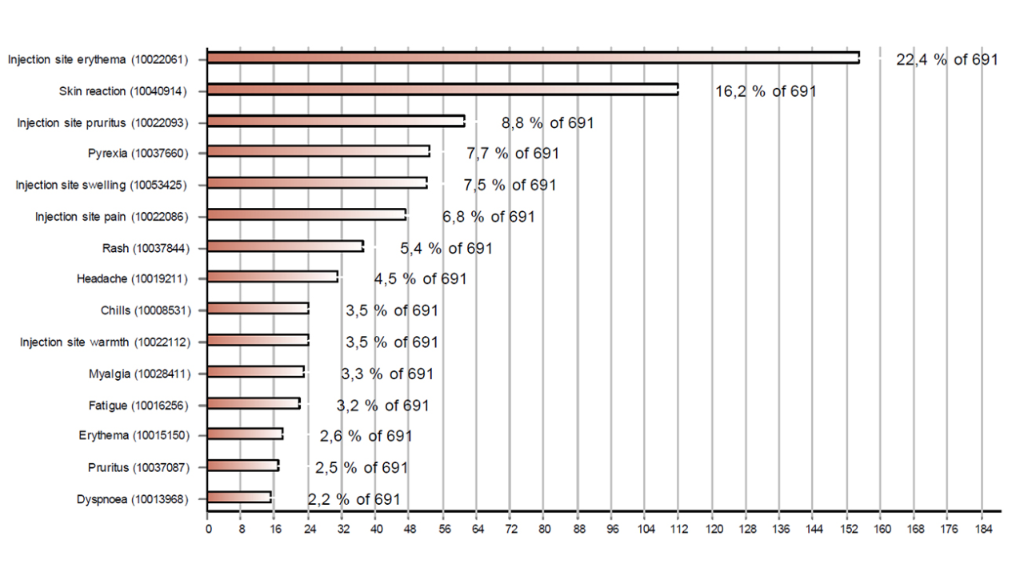
Figures 1 and 2 respectively show the 15 most frequently reported reactions associated with Comirnaty® and Moderna's COVID-19 vaccine. The most common reactions were injection site reactions (redness, itching, pain and swelling), fever, headaches, muscle pain and shivering. Cases with these reactions accounted for more than half of the reports (56%).
In 37 serious cases, the people concerned died at differing intervals after receiving the vaccine. Their average age was 84 years, and the majority of them had serious pre-existing conditions. These cases were analysed with particular care. As far as is known at present, death was caused by conditions such as infections, cardiovascular events or diseases of the lungs and airways that occurred independently of the vaccinations. Despite a chronological correlation, there is no concrete evidence to suggest that the vaccination was the cause of death. The drug regulatory authorities of other countries and the WHO reached similar conclusions when they evaluated deaths with a temporal link to the COVID-19 vaccines authorised in Switzerland.
The reports of adverse reactions received and analysed to date do not change the positive benefit-risk profile of the COVID-19 vaccines. The reports now provide us with a clearer picture of the vaccines’ safety in daily use. This largely confirms their known side effects profile. Known side effects of COVID-19 vaccines are listed in the continually updated medicinal product information texts published on www.swissmedicinfo.ch.
Information on reporting ADR associated with COVID-19 vaccines
Please note: Known, non-serious reactions are not subject to the reporting obligation prescribed by Art. 59 of the Therapeutic Products Act. The known, non-serious and very common reactions to COVID-19 vaccines include transitory pain and swelling at the injection site, fatigue, shivering, fever, headache and muscle and joint pain. These passing local and general reactions are normally a sign of the body dealing with the vaccine.
However, serious or as yet unknown adverse reactions must be reported. At this relatively early stage of the vaccination campaign, doctors should generally report all ADR that they feel are medically relevant. For this purpose they should use the ElViS electronic reporting tool, which they also can log into using their HIN ID.
ElViS has now been further simplified specifically for reports involving COVID-19 vaccines: after log-in, clicking a red button leads directly to a specific report form. When the respective vaccine is selected in this form, some of the fields are prefilled automatically in order to further simplify the reporting process.
1) These reports relate to cases of COVID-19 where there was a correlation in time with the vaccination. Since the vaccinated persons had not been tested before they were vaccinated and the human body first needs to build up its protection against the virus, the onset of COVID-19 disease in temporal correlation with vaccination is possible. However, there is no causal link with the vaccination. Unlike live vaccines – e.g. those against measles, mumps and rubella (MMR) – COVID-19 vaccines do not contain (attenuated) viruses and cannot therefore trigger COVID-19 disease.