Swissmedic is responsible for monitoring the safety of medicinal products. This involves receiving and evaluating reports on side effects of medicines and vaccines, including COVID vaccines. However, Swissmedic does not answer questions about individual therapies or vaccination issues, nor does it make any recommendations regarding them. For such enquiries, please contact your doctor.
Under the Swiss Therapeutic Products Act, consumers, patients and their organisations and interested third parties can report adverse drug reactions and incidents to Swissmedic.
Please be aware that you should still talk to your doctor or chemist if you are worried about suspected adverse reactions or an incident.
Reporting a general adverse reaction to a medicinal product or a suspected adverse reaction in connection with a COVID-19 vaccine
Private individuals can now submit all reports of suspected adverse reactions to human medicinal products to Swissmedic via a web form. You can report an adverse drug reaction for yourself or on behalf of another person, such as a child or relative.
Important information on reports by private individuals
Please note that reporting a side effect does not replace either a medical examination or a diagnosis. Although there may be a temporal relationship between the administration of a medicinal product or vaccine and the onset of a symptom or illness in an individual case, this does not necessarily mean that the medicinal product or vaccine was actually the cause. Other possible causes, which may possibly require targeted therapeutic measures, can only be excluded with a medical diagnosis.
If you notice symptoms that could be a side effect, you should first contact the place that prescribed, dispensed or administered the medicinal product. Always consult your treating physician in the event of symptoms/ailments. Healthcare professionals can assess whether this involves a side effect that must be reported or symptoms that are more likely to be caused by something else. You can potentially ask your healthcare professional (doctor, pharmacist) to report the side effect.
By reporting side effects or suspected cases, you are making an important contribution to safe medicinal products and vaccines. However, please note that Swissmedic is unable to answer individual treatment or health questions and cannot give any corresponding recommendations.
General Information
FAQ
Need for pharmacovigilance
Medicines are developed over a period of several years. The efficacy and safety of a new drug are generally studied on a few thousand carefully selected and followed-up trial subjects and patients according to strictly defined criteria. For this reason only very frequent adverse reactions – mainly those depending on the drug's pharmacological properties – can be observed during its clinical development.
Once the product has been placed on the market, a much larger – and also often polymorbid – population will be exposed, which may lead to a change in the drug's hitherto known safety profile. Adverse drug reactions can then be observed more frequently, including those occurring only sporadically and independently of the pharmacological properties of the substance. These adverse reactions of drug treatment observed in daily practice must be reported without delay. If such information is consistently forwarded to the Swissmedic National Pharmacovigilance Centre, hitherto unknown risks can be identified and tackled.
The Swissmedic National Pharmacovigilance Centre accepts and processes reports of adverse drug reactions. The National Pharmacovigilance Centre is supported by five regional centres that are each affiliated to a university department; in particular, these process reports from healthcare professionals that involve an important safety signal. For its part, the pharmaceutical industry forwards reports of adverse reactions that it receives to Swissmedic. The National Pharmacovigilance Centre works closely with the World Health Organization (WHO) international centre for drug safety.
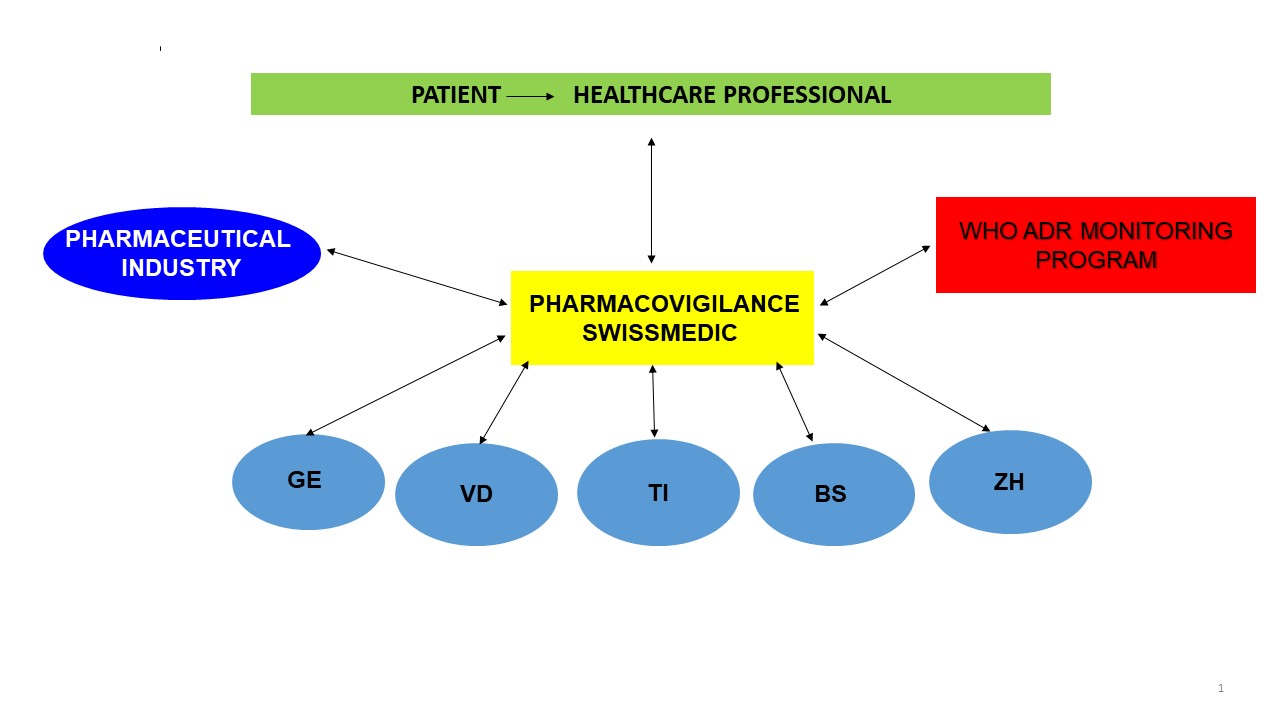
In accordance with the Therapeutic Products Act, adverse reactions that are serious or unknown, or that are inadequately described in the information for healthcare professionals for the medicinal product in question, must be reported.
Adverse reactions are considered serious if they:
- result in death
- are life-threatening
- lead to, or prolong, hospitalisation
- involve a persistent disability or incapacity
- are otherwise to be considered medically significant (if, for example, a timely medical intervention prevented one of the above-mentioned outcomes)
These suspected adverse reactions should be reported within 15 days of diagnosis; non-serious reactions should be reported within 60 days.
A causality between a reaction and a medicine need not be proved: suspicion alone is sufficient to justify reporting such events.
Although misuse, dependency and addiction are not covered by the WHO definition of an adverse drug reaction, as they do not relate to normal posology, it is important to report such events as they might affect the safety profile of a drug.
Under the Therapeutic Products Act, all professionals who are entitled to distribute, administer or prescribe medicines are obliged to report suspected adverse drug reactions. Pharmaceutical companies that manufacture or distribute ready-to-use medicinal products are also subject to a reporting obligation.
However, patients may also report undesirable side-effects of drug therapy. A consultation with the family doctor leading to a joint report has the advantage of providing relevant medical details such as results of investigations, but this is not compulsory.
Private individuals
Patients, relatives and interested third parties can now also report suspected side effects and incidents electronically to Swissmedic without the need for any medical expertise. Receipt of the report is confirmed automatically in the application. No additional confirmation receipt is sent by e-mail.
Healthcare professionals
Electronic reporting via the ElViS portal is the preferred route for healthcare professionals. ElViS can also be used to submit case-related documents, such as laboratory reports or hospital discharge reports.
Companies
Marketing authorisation holders can submit suspected adverse drug reactions either via the ElViS portal (Electronic Vigilance System) or via a gateway for the electronic exchange of individual case safety reports.
The Swissmedic pharmacovigilance team carefully screens all incoming reports for new risks. If a new risk is identified, the need for action is evaluated and appropriate measures are taken in collaboration with the Swissmedic department responsible for the drug concerned.
Support/Contact
Address
Swissmedic
Pharmacovigilance unit
Hallerstrasse 7
3012 Bern (Switzerland)
Note on enquiries
Please note that Swissmedic is not able to answer any questions on individual treatment or vaccination and cannot give any related recommendations. Please always contact your doctor for diagnosis and treatment of your symptoms or complaints.
Last modification 15.12.2025

