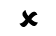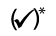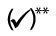What do users need to know?
Procurement:
Under the transitional provisions in Art. 106 para. 2 to 6 MedDO, it will be possible to procure devices in the product groups listed in Annex 1 MedDO without any restriction until 1 May 2024 under the existing sector-specific legislation.
Swissmedic recommends that a check for conformity with the relevant regulatory requirements is included during the procurement process and that documentation on product conformity (declaration of conformity and EC certificates) is checked and kept.
Information and help on the procurement can be found on our website:
Reporting incidents & FSCAs (vigilance)
Professionals who use devices have a legal obligation to report serious incidents to Swissmedic.
More information is available at:
With regard to monitoring of use, and for questions regarding whether uses are permitted, please contact the Cantonal Department of Health.
The cantons are responsible for monitoring retail trade and dispensing points (Association of Cantonal Pharmacists).
Treatments
Users must know which treatments they are permitted to perform The table below provides a convenient overview of the authorisation rules according to product group:
Legend:
✓ = permitted;
(✓) = permitted under medical supervision;
✗ = not permitted
* Qualified healthcare professionals under direct supervision of a doctor (Annex 6 MedDO)
**For treatments using non-ionising radiation and sound, the Federal Act on Protection against the Risks associated with Non-Ionising Radiation and with Sound (NIRSA) must be observed. The act and ordinance entered into force on 1 June 2019.
Effective 1 June 2024, the treatments set forth in the Ordinance on Protection against the Risks associated with Non-Ionising Radiation and with Sound (O-NIRSA) can be performed only by persons with a certificate of competence. Please refer to the FOPH website for further information: Treatments using non-ionising radiation and sound (admin.ch).
***Devices for personal use for depilation in cases where excess hair is not due to an illness